US study shows AstraZeneca vaccine safe and effective: Should encourage Malawians to get jab
AstraZeneca’s Covid-19 vaccine being administered in Malawi developed with Oxford University is is both safe and highly effective, new trial data from the United States shows.
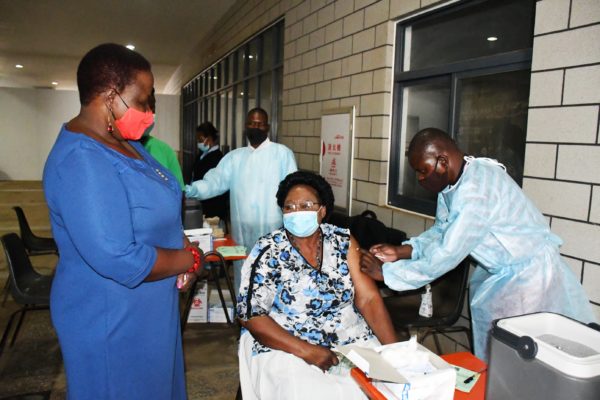
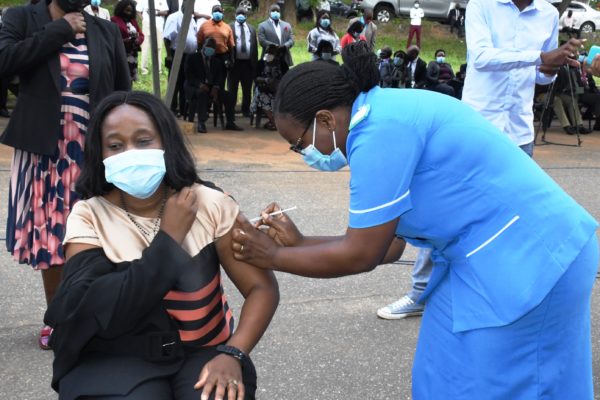
Research involving more than 30,000 participants revealed the jab was 79 per cent effective at preventing symptomatic illness, 100% effective against severe or critical disease and hospitalisation, according to the U.S.-based Phase 3 study.
Data showed “no safety concerns related to the vaccine,” said AstraZeneca.
Malawi health experts said the new study and the seal of approval by World Health Organisation (WHO) on the safety of the Oxford/ AstraZeneca vaccine should encourage all Malawians to receive the vaccine once they get a chance.
Society of Medical Doctors in Malawi president Dr Victor Mithi said they have been monitoring some people after getting the vaccine, especially health workers and no reports so far of blood clotting have been reported.
He admitted there are still some people in the country who believe the misconceptions about clotting, even though such issues have been cleared.
Ministry of Health Principal Secretary Dr Charles Mwansambo said many Malawians were keen to get the jab.
He urged all stakeholders, including the media, to rally people and raise awareness on the vaccine by sharing information “about Covid-19 and the vaccine.”
Mwansambo also said about 100 000 more doses from the African Union will arrive in the country soon while an extra 1.26 million doses from the Covax facility will be in by end of May.
Chakwera launched the Covid-19 vaccination on March 11 alongside other leaders, including Vice-President Saulos Chilima, former presidents Joyce Banda and Bakili Muluzi and other leaders with an appeal to Malawians to get the jab.


Kutsogoloku wodwala Covid-19 will be treated like we treated wa nthomba as per Maliro ndi Miyambo ya Chewa. Amene mukukana vaccine like the false prophets Kambale and Mbewe.
Mmalo moti mudzitiuza kuti Dr. Chidongo Bwangazu ndi Dr. Kwampenya Mpokoni a ku College of Medicine apanga vaccine okomera a malawi mukukatitchulira azungu? Ukapolo utha koma mochedwa
What’s next after that? We need more for the remaining pipo before cold weather starts.
Chonde musaiwalr Petulo ku Mngochi mukamulase naye angakuneneni